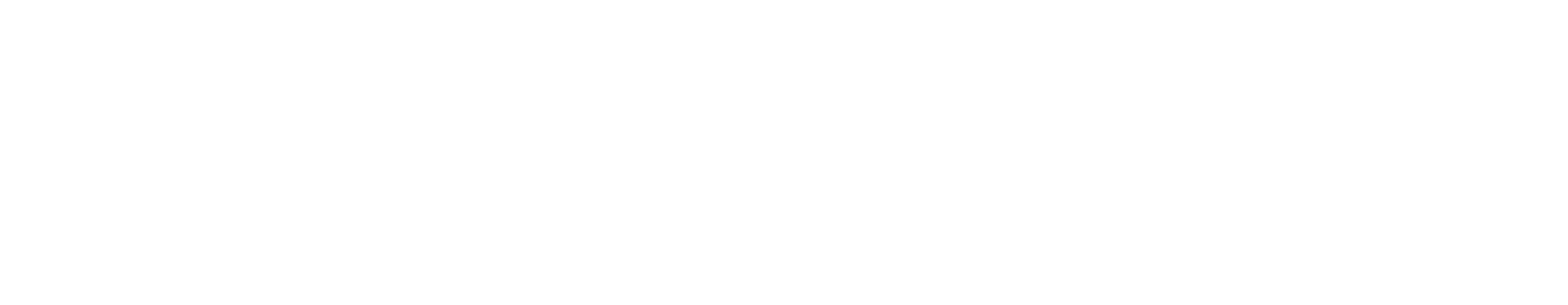Amgen’s Investigational Bemarituzumab Receives Breakthrough Therapy Designation
The FDA granted breakthrough therapy designation to an experimental drug from Amgen for patients with FGFR2b over expressing and HER2-negative gastric cancer.
Basilea starts phase 1/2 study FIDES-03 with derazantinib in patients with gastric cancer
Basilea Pharmaceutica has started a study of a drug that inhibits the cancer-driving protein FGFR. It will be studied alone and with... Read More
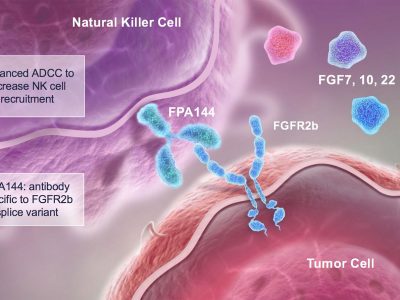
Enhertu now approved in Japan for gastric cancer
Enhertu from Daiichi-Sankyo is now approved in Japan for the treatment of HER2-positive gastric cancer.
Leap Therapeutics and BeiGene Announce First Patient Dosed in Study of DKN-01 in Combination with Tislelizumab for the Treatment of Metastatic Gastric or Gastroesophageal Junction Cancer
Leap Therapeutics and BeiGene have started dosing patients in a trial combining an anti-PD-1 antibody with the novel targeted drug DKN-01 in... Read More
Ramucirumab and durvalumab for previously treated, advanced non–small-cell lung cancer, gastric/gastro-oesophageal junction adenocarcinoma, or hepatocellular carcinoma
Combining Cyramza with IMFINZI® (durvalumab) was safe and effective in a small trial that included gastric cancer patients.
Pieris and Lilly enter into a Clinical Trial Collaboration to Evaluate Combination of PRS-343 with Ramucirumab and Paclitaxel in Gastric Cancer
Pieris and Eli Lilly have partnered to test a combination of Cyramza, paclitaxel and Pieris’ novel bispecific antibody in HER2-positive gastric cancer.
MacroGenics Announces Margetuximab Granted Orphan Drug Designation in the U.S. for Gastric Cancer
MacroGenics received orphan drug designation from the FDA for its investigational gastric cancer drug margetuximab.
First-Line Pembrolizumab and Trastuzumab in HER2-Positive Esophageal, Gastric, or Gastroesophageal Junction Cancer
Patients with Her2-positive gastric cancer taking a combination of the immune-boosting drug Keytruda with Herceptin had progression-free survival rate at six months... Read More
Regorafenib Plus Nivolumab in Patients With Advanced Gastric or Colorectal Cancer
A recent trial of a combination of Stivarga and Opdivo showed better activity than either drug alone in patients with gastric cancer.
FDA Grants Full Approval of Deciphera Pharmaceuticals’ QINLOCK™ (ripretinib) for the Treatment of Fourth-Line Gastrointestinal Stromal Tumor
The FDA approved Deciphera Pharmaceuticals’ Quinlock (ripretinib) for the treatment of advanced gastrointestinal stromal tumors.